BC99 Clinical Research——Sports Nutrition
As of February 2026, more than 1,000 subjectsacross all age groups-including children, adults, andthe elderly-have been enrolled in clinical trialsinvolving BC99. Among the clinical interventionregimens, the maximum daily dosage administeredwas 48 billion CFU, with the longest interventionduration lasting up to 12 weeks.No serious adverse events (SAEs) associated withBC99 were reported in any dosage group or agesubgroup throughout the entire clinical trial period.No abnormal alterations were observed in laboratorytest parameters (covering complete blood count,hepatic and renal function tests, etc.), vital signs, orroutine physical examinations.Collectively, these robust findings verify that BC99possesses an excellent safety and tolerance inhumans.
BC99 Promotes Protein Digestion and Metabolism in Active Individuals
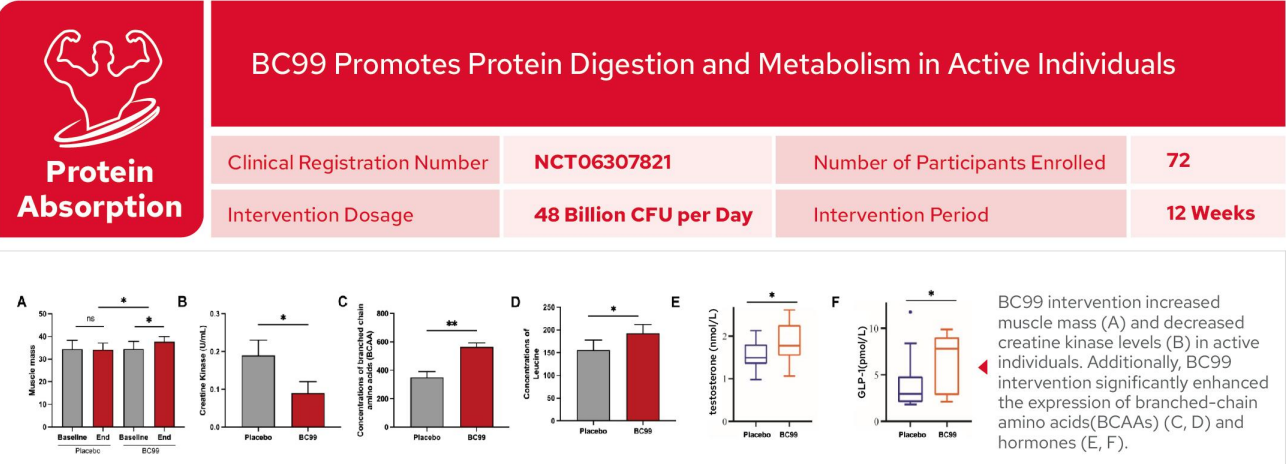
Following BC99 intervention,muscle mass was significantly augmented and exercise-induced muscle damage was mitigated in physically active individuals.BC99 intervention markedly upregulated the expression of BCAAs,including leucine,alongside
elevated levels of testosterone and glucagon-like peptide-1(GLP-1).These findings demonstrate that BC99 not only promotes increases in muscle mass but also facilitates robust protein digestion and metabolic turnover






 Leave a Message
Leave a Message Email
Email Linkedin
Linkedin