BC99 Clinical Research——Mental Health
As of February 2026, more than 1,000 subjectsacross all age groups-including children, adults, andthe elderly-have been enrolled in clinical trialsinvolving BC99. Among the clinical interventionregimens, the maximum daily dosage administeredwas 48 billion CFU, with the longest interventionduration lasting up to 12 weeks.No serious adverse events (SAEs) associated withBC99 were reported in any dosage group or agesubgroup throughout the entire clinical trial period.No abnormal alterations were observed in laboratorytest parameters (covering complete blood count,hepatic and renal function tests, etc.), vital signs, orroutine physical examinations.Collectively, these robust findings verify that BC99possesses an excellent safety and tolerance inhumans.
Improvement of Anxiety and Depression in Adults by BC99
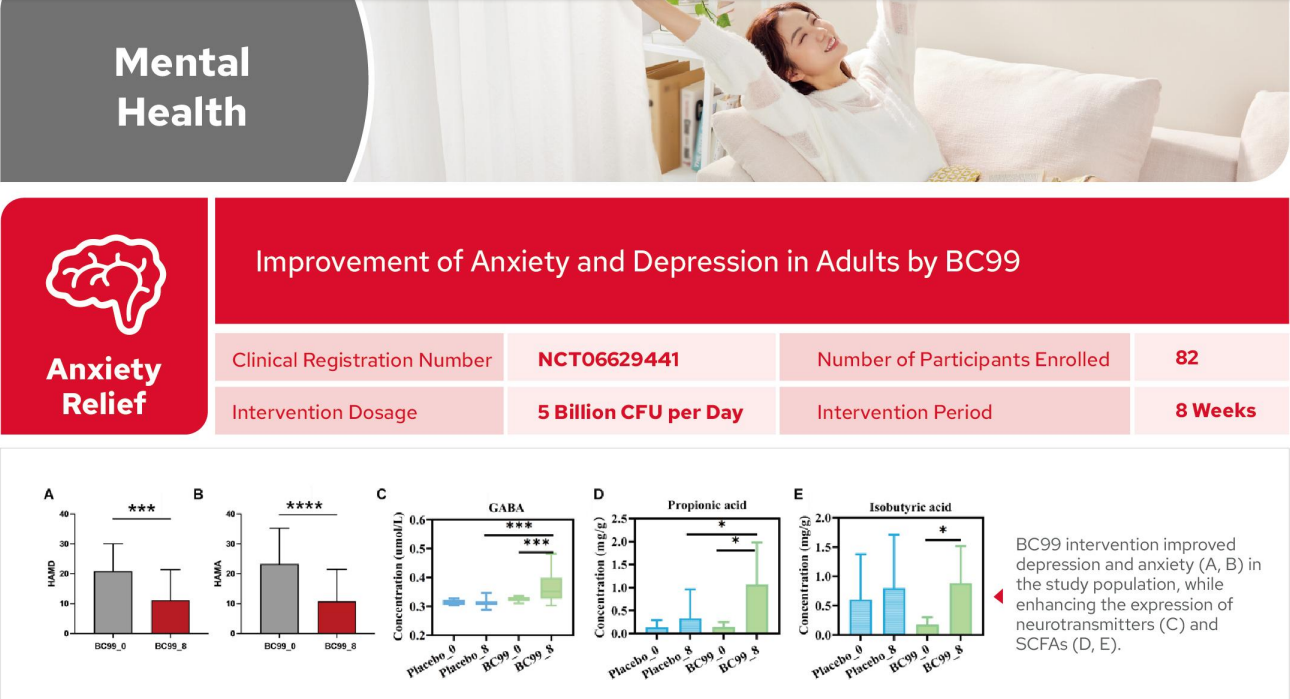
Following BC99 intervention,significant reductions were observed in both the Hamilton Depression Rating Scale(HAMD)and
Hamiton Anxiety Rating Scale(HAMA)scores in the study population,accompanied by a marked increase in GABA expression.These findings indicate that BC99 regulates neurotransmitters to alleviate anxiety and depression.
Additionally,BC99 intervention significantly enhanced the expression of propionic acid and isobutyric acid,thereby influencing
neuronal function.This suggests that BC99 confers emotional health benefits to the study population by increasing SCFAs levels





 Leave a Message
Leave a Message Email
Email Linkedin
Linkedin