Scalable Intelligent Biomanufacturing of BC99
Industrialization of Probiotics
Following more than a decade of industrial research,BC99 has successfully transitioned from laboratory-scale production to
large-scale manufacturing.Through continuous process optimization,the technology has consistently overcome technical barriers. The production process employs advanced fermentation technologies and a rigorous quality control system,ensuring that each
batch of the product possesses stable and highly effective probiotic properties.
Currently,BC99 can provide probiotics powder with a specification of 300 Billion CFU/g tailored to application needs. The product demonstrates excellent stability and is free from harmful substances such as melamine,aflatoxins,and plasticizers,meeting the requirements for healthy foods.
Furthermore,to meet the demands of diverse markets,BC99 has been developed into various dosage forms,including capsules, powders,and tablets,providing consumers with the flexibility to choose a suitable administration method based on their individual needs and preferences.

Through the application of a 0.1mL-6T stepwise scale-up validation and post-processing industrial technology
platform,BC99 and other strains have successfully achieved seamless transition from laboratory to industrial
production,accompanied by continuous process engineering optimization.During the production phase,the BC99 manufacturing line is equipped with high-precision detection instruments that enable real-time monitoring of critical fermentation parameters-including pH,dissolved oxygen concentration,and nutrient consumption-thereby ensur- ing a stable and controlled production environment.Furthermore,refined management practices have been imple- mented across all production stages,including raw material selection,fermentation control,drying processes,and
packaging,further enhancing the purity and activity of the final product.
WecSimLarge-Scale Intelligent Manufacturing of Microbial Strains

Employing an intelligent control system,the entire production process of BC99 is carried out in a GMP-compliant constant
temperature and humidity purification workshop that meets pharmaceutical-grade standards.A 200-ton fermentation system efficiently achieves high-density cultivation of the bacterial cells,which are subsequently subjected to fine processing through multi-stage centrifugal separation technology.The bacterial cells are ultimately transformed into a high-quality bacterial powder product via a spray-drying system.
Testing Methods

An independent testing center has established a comprehensive,multi-dimensional quality control system for probiotics,
enabling precise monitoring of strain counts,purity identification,verification of authentic strain addition,and prevention of non-target microbial contamination.
Stability Evaluation
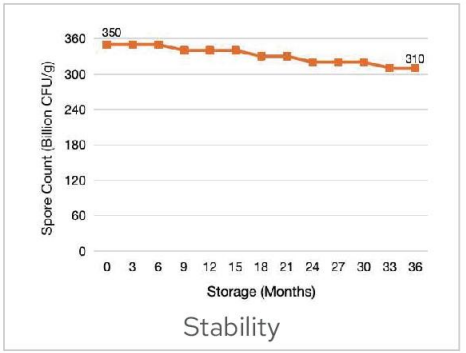
Stability
BC99 probiotics powder can be stored under ambient conditions,with a shelf life of 36 months from the date of manufacture.In accordance with the ICH guidelines on climatic zone stability,stability assessments were conducted on BC99 products,and the test results confirmed compliance with product quality requirements
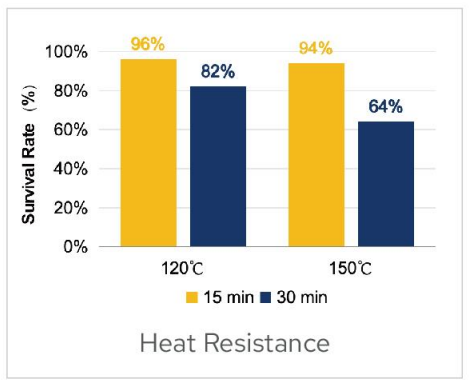
Heat Resistance
Heat resistance testing was conducted on BC99 probiotics powder.The results demonstrated that BC99 can tolerate temperatures as high as 150℃.After
treatment at 150℃ for 30 minutes,the survival rate remained at 64%.




 Leave a Message
Leave a Message Email
Email Linkedin
Linkedin