Industry Insights
Home > News > Industry Insight > From Gut to Immune-Skin Health: LRa05 Clinical Trial Brings New Hope for Children with Atopic Dermatitis (NCT05989295)
From Gut to Immune-Skin Health: LRa05 Clinical Trial Brings New Hope for Children with Atopic Dermatitis (NCT05989295)

Atopic Dermatitis: An Immune-Related Challenge
Atopic dermatitis (AD) affects up to 20–25% of children worldwide, with preschool prevalence reaching 16–18%. Children with AD often experience around nine flare-ups per year, characterized by intense itching, dry skin, and disrupted sleep, which also impose a substantial emotional and practical burden on parents.
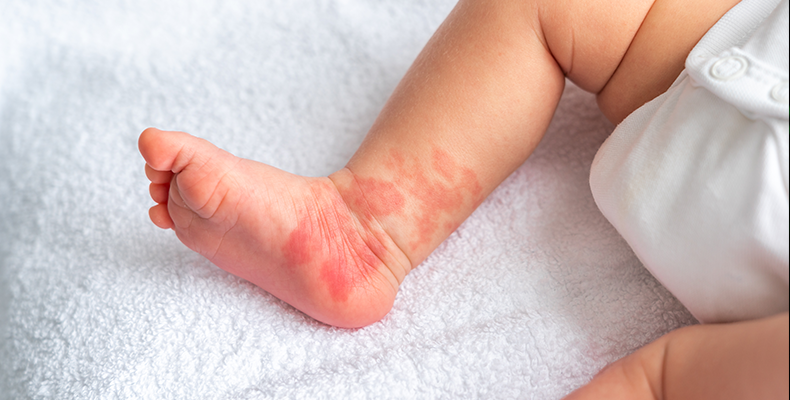
While topical corticosteroids remain standard treatment, long-term use is limited by side effects and relapses. Emerging evidence links gut microbiota imbalances to AD, suggesting that probiotics may help restore microbial balance and support immune and skin health. To explore this, a randomized, double-blind, placebo-controlled trial (NCT05989295) was conducted in children with AD.
The Clinical Trial: Design and Approach
• Participants: 32 children aged 2–14 years with AD (SCORAD 16-40)
• Study duration: 12 weeks
•Intervention: LRa05 group received 2B CFU/sachet daily; placebo group received maltodextrin (2g/sachet)
• Key measures: Eczema symptoms (SCORAD index), skin hydration, and safety
Key Findings
• Safety: LRa05 was well tolerated, with no adverse events reported.
• Decreased Eczema Outbreaks: LRa05 supplementation led to a 30% reduction in outbreak frequency.
• Skin Barrier Support: Children in the LRa05 group showed an 8.5% increase in skin hydration (p < 0.05).
Overall, statistically significant benefits were observed in children with mild AD. The full study has been submitted for peer-reviewed publication, and detailed results will be made available upon publication.
Wecare Delivering Clinically Validated Solutions
The clinical validation of LRa05 highlights its potential as a safe, natural, and scientifically supported solution for children with immune-related skin conditions. This milestone reinforces Wecare’s commitment to translating clinical research into meaningful health outcomes.
Our mission drives us forward: Quality Probiotics, Quality Life.
Learn more:【 http://www.wecare-life.com】
Contact Us: 【 sales@wecare-life.com】








 Leave a Message
Leave a Message Email
Email Linkedin
Linkedin